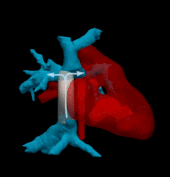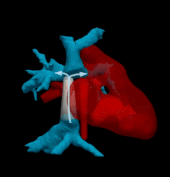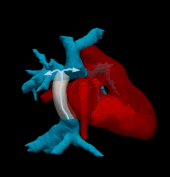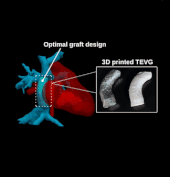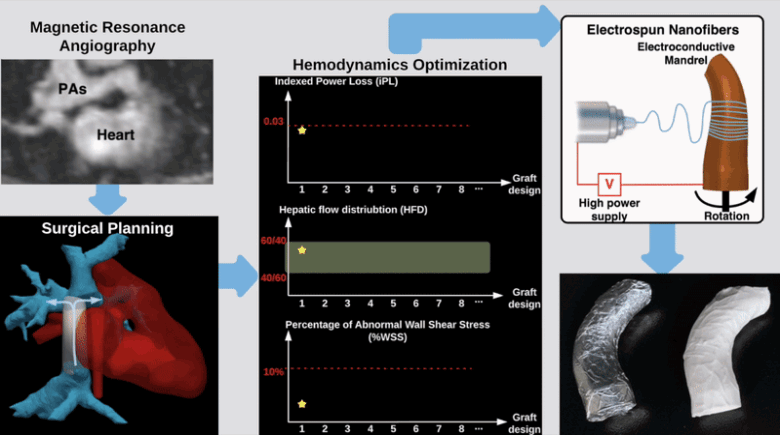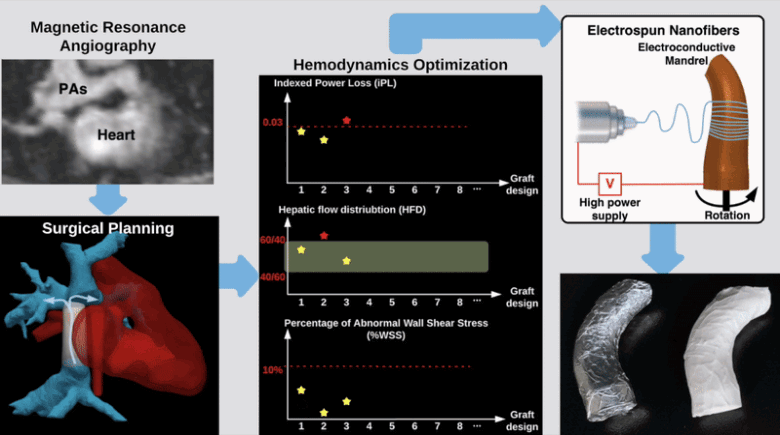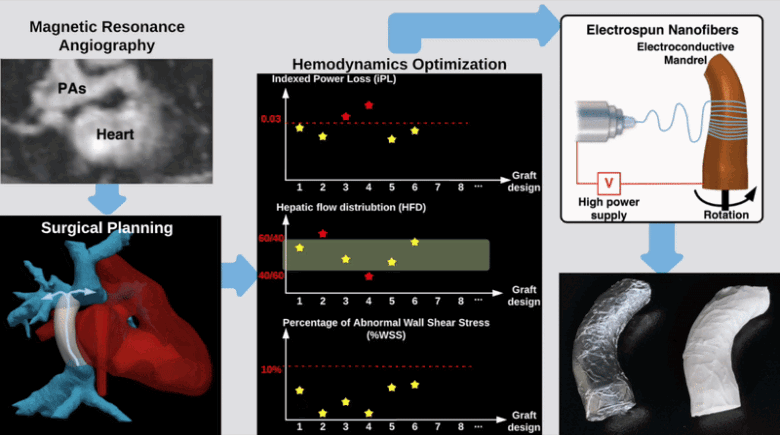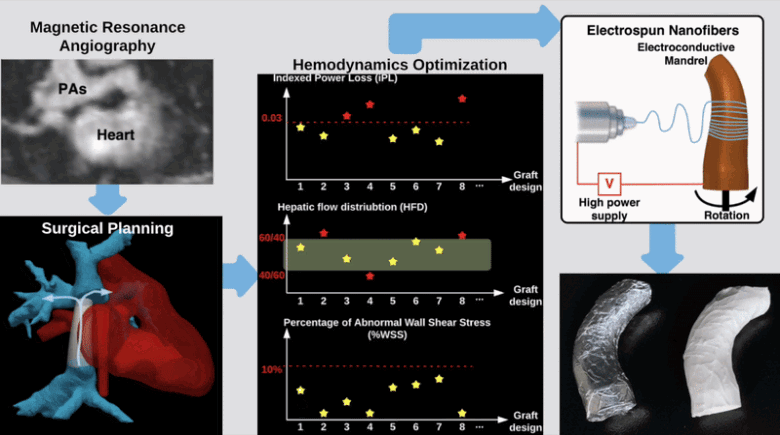
Hemodynamically optimized tissue engineered vascular grafts could reduce post-operative complication and provide long-term health benefits for patients undergone Fontan surgery, which is the hallmark operation in the surgical management of single ventricle congenital heart disease patients. Designing patient-specific Fontan grafts is a complex task due to the variety of patient-specific anatomies, confined surgical planning space, and the requirement of simultaneously considering multiple criteria for graft design optimization. This work aims to develop a semi-automatic Fontan surgery planning method for designing and manufacturing hemodynamically optimized patient-specific grafts. To achieve this goal, we parameterized Fontan grafts to explore patient-specific vascular graft design spaces and search for optimal solutions by formulating a nonlinear constrained optimization problem, which minimizes indexed power loss (iPL) of the Fontan model by constraining hepatic flow distribution (HFD), percentage of abnormal wall shear stress (%WSS) and geometric interference between Fontan grafts and the heart models within clinically acceptable thresholds.
We tested the proposed method on two patient-specific models (n=2). Our results demonstrated that the automatically optimized (AutoOpt) Fontan models hemodynamically can outperform or at least are comparable to manually optimized Fontan models with significantly reduced surgical planning time (15 hours versus over 2 weeks). We also demonstrated feasibility of manufacturing the AutoOpt Fontan grafts by using electrospun nanofibers. Our technique demonstrated great potential to bridge the gap between machine intelligence and clinical medicine, allowing the surgeon to directly incorporate their unique understanding of surgical field into the design of Fontan grafts, as well as directly receiving automatically optimized surgical planning based on the surgeon’s preferred Fontan pathways.