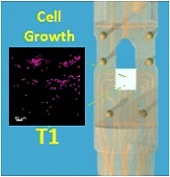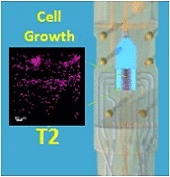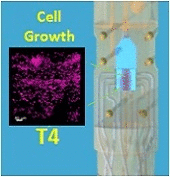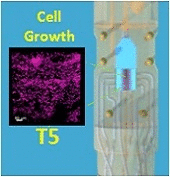
The development of stable, functional tissue is a challenge in regenerative medicine. Tissue engineering (TE) approaches that involve seeding cells into pre-determined tissue scaffolds ignore the complex environment where the material properties are spatially inhomogeneous and evolve over time. The article presents a novel methodology for spatiotemporal control of mechanical forces in scaffolds that enables generating on demand flow fields in real-time and can adapt to changes in the environment.
The flexibility of dialing-in arbitrary shear stress distributions and adjusting flow field patterns in a scaffold over time in response to cell growth was achieved with a multi-inlet bioreactor and a control algorithm with learning capabilities to dynamically solve the inverse problem of computing the inlet pressure distribution required over the multiple inlets to obtain a target flow field. By adjusting the pressures at each inlet we can produce arbitrary flow fields, generating various mechanical force distributions within a single scaffold.
Real-time control can be achieved by employing this flow control strategy as part of a feedback control loop that is guided by non-invasive imaging. The bioreactor is made of non-magnetic material for compatibility with a nuclear magnetic resonance (NMR) environment where interstitial flows in the porous scaffold matrix inside the reactor are periodically measured. The measurements are used for estimating changes in the hydrodynamic properties of the scaffold to update the control algorithm which finds the appropriate inlet pressure to be dialed-in at each inlet, thereby offering flexible means of dynamic control that can adapt to structural changes.
Our proposed methodology constitutes a new platform for studies of cellular responses to mechanical forces in complex environments and opens potentially transformative possibilities for TE.
Keywords: shear stress, bioreactor, porous scaffold, flow control, tissue engineering