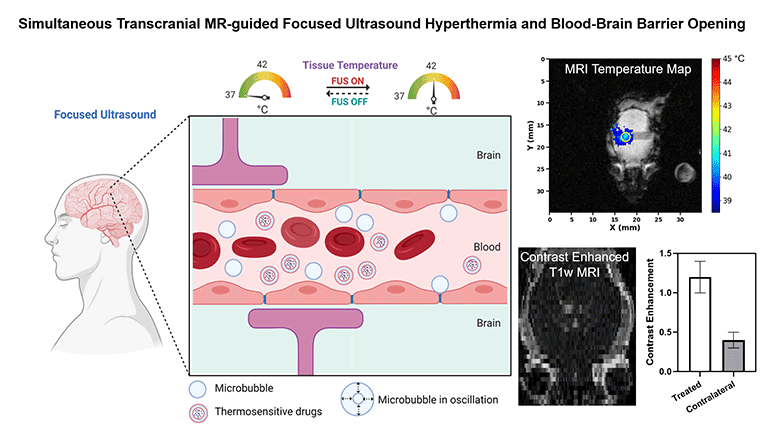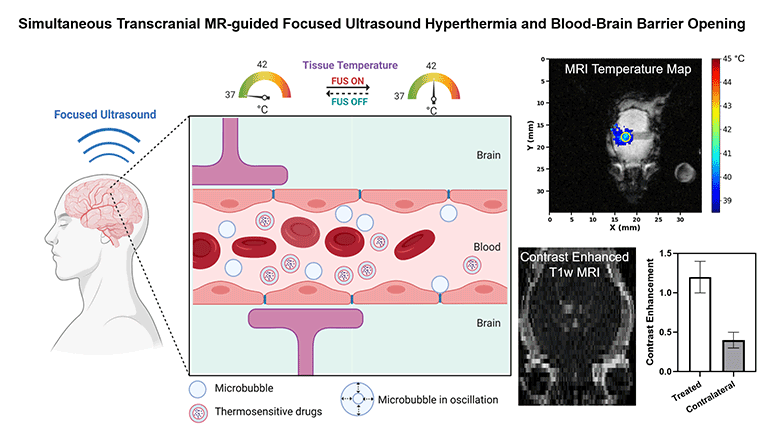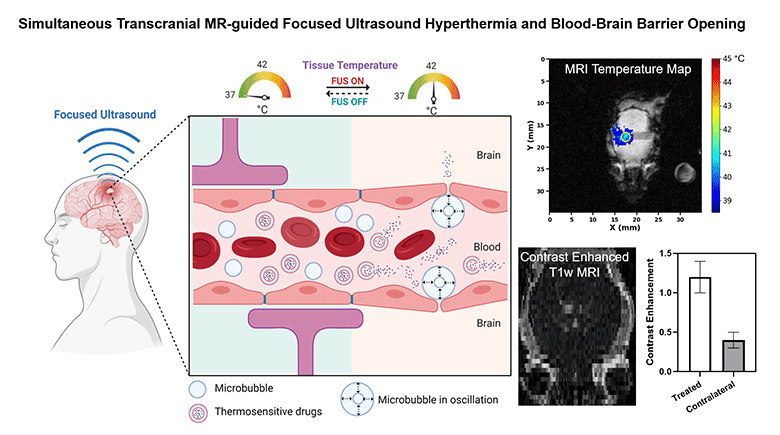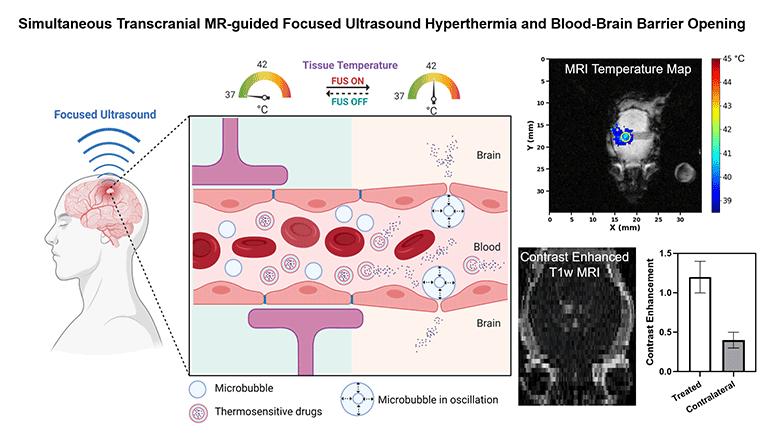
Non-invasive methods to enhance targeted drug delivery in the brain have been pursued for decades. We present a synergistic focused ultrasound-based brain drug delivery method for both spatiotemporally precise mild hyperthermia and simultaneous localized blood-brain barrier (BBB) opening. We hypothesized that the presence of microbubbles (MBs) combined with MR-guided focused ultrasound (MRgFUS) can reduce the ultrasound power required for transcranial hyperthermia, thus improving the safety profile of this technique, and can simultaneously open the BBB locally, thereby enhancing the drug penetration synergistically.
To demonstrate the feasibility and safety of the proposed method, we performed transcranial hyperthermia (42 °C, 10 min) in wild-type mice using a small animal MRgFUS system incorporated into a 9.4T Bruker MR scanner, with infusions of saline or Definity® MBs with different doses. MR thermometry data was continuously acquired as feedback for the ultrasound controller during the experiment. BBB opening was evaluated with contrast-enhanced T1-weighted MR imaging. Safety was assessed with T2-weighted MR imaging and histology.
We demonstrated that spatiotemporally precise transcranial hyperthermia was achieved in both saline and MB groups. A significant ultrasound power reduction (-45.7%, p=0.006) was observed in the MB group with a dose of 20 µl/kg/min compared to saline. Localized BBB opening was achieved in MB groups confirmed by contrast-enhanced T1-weighted MR images. No structural abnormality, edema, hemorrhage, or acute microglial activation was observed in the target region from T2-weighted MR imaging, H&E staining, and immunohistochemistry. Our investigations showed that it is feasible and safe to achieve localized transcranial hyperthermia at significantly reduced powers and to simultaneously open the BBB via transcranial MRgFUS and MBs. This study provides a new synergistic strategy for enhanced drug delivery in brain tissues with a high clinical translation potential.
