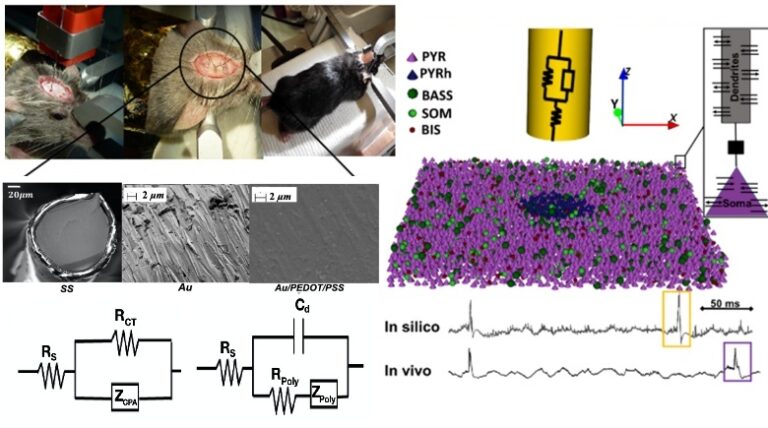
Microelectrodes allow for the recording of neural activities with a high spatial resolution. However, their small sizes result in high impedance causing high thermal noise and poor signal-to-noise ratio. In drug-resistant epilepsy, the accurate detection of Fast Ripples (FRs; 250-600 Hz) can help in the identification of epileptogenic networks and Seizure Onset Zone (SOZ). Consequently, good-quality recordings are instrumental to improve surgical outcome. In this work, we propose a novel model-based approach for the design of microelectrodes optimized for FRs recording.
A 3D microscale computational model was developed to simulate FRs generated in the hippocampus (CA1 subfield). It was coupled with a model of the Electrode-Tissue Interface (ETI) that accounts for the biophysical properties of the intracortical microelectrode. This hybrid model was used to analyze the microelectrode geometrical (diameter, position, and direction) and physical (materials, coating) characteristics and their impact on recorded FRs. For model validation, experimental signals (local field potentials, LFPs) were recorded from CA1 using different electrode materials: stainless steel (SS), gold (Au) and Au coated with poly(3,4-ethylene dioxythiophene) /Poly(styrene sulfonate) (Au:PEDOT/PSS).
Our study indicated that a radius between 65-120 µm radius wire microelectrode are the most optimal for recording FRs. In addition, in silico and in vivo quantified results showed a possible improvement in FRs observability using PEDOT/PSS coated microelectrodes. According to our work, the optimization of the design of microelectrodes for FRs recording can improve the observability and detectability of FRs which are a recognized marker of epileptogenicity. This original model-based approach presented in this work can assist in the design of hybrid electrodes that are optimized for presurgical investigation of the SOZ and can potentially improve the outcomes of resection surgery for epileptic patients with drug-resistant epilepsy.