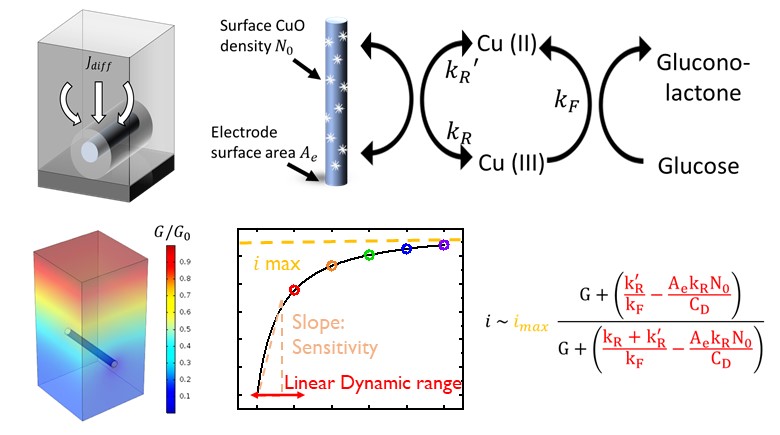
For the 422 million diabetes patients worldwide, the glucose sensor is a life-saving technology. The first-generation sensors use glucose oxidase (GO_x) enzyme to convert glucose into gluconic acid and hydrogen peroxide (H_2 O_2). The amperometric response of H_2 O_2 determines the blood glucose level. Although most glucose meters in the market are still enzyme-based, the intrinsic instability of GO_x makes integration onto wearable and implantable platform difficult. First, GO_x activity depends sensitively on temperature. The degree of temperature control needed is incompatible with long-term bio-implantable applications. Second, the consumption of O_2 during glucose oxidation causes the sensor reading to drift, requiring frequent calibration. Finally, the reaction by-product H_2 O_2 is potentially harmful to the human body.
The development of a non-enzymatic electrode for glucose detection promises a new generation of glucose sensors. Over the past decade, the development of nanostructured material has enabled metal-oxide based non-enzymatic glucose sensing with consistently high sensitivity, making it of considerable interest for future always-on wearable closed-loop sensing for hypoglycemia management. In this work, we develop a physics-based model to correlate the geometrical and chemical design parameters to the non-linear amperometric response of non-enzymatic glucose sensor on the geometrically complex substrate. Using this model, we can interpret the scattered results in the literature within a common conceptual framework. Our results show that while non-enzymatic glucose sensor still does not have sufficient dynamic range to replace the classical blood glucose sensors, these sensors could be useful for low concentration glucose sensing applications involving sweat, saliva, and ocular fluid. Our model will predictably improve the design of non-enzymatic glucose sensors for integrating into a continuous glucose monitoring system (CGMS) in wearable and implantable platforms.