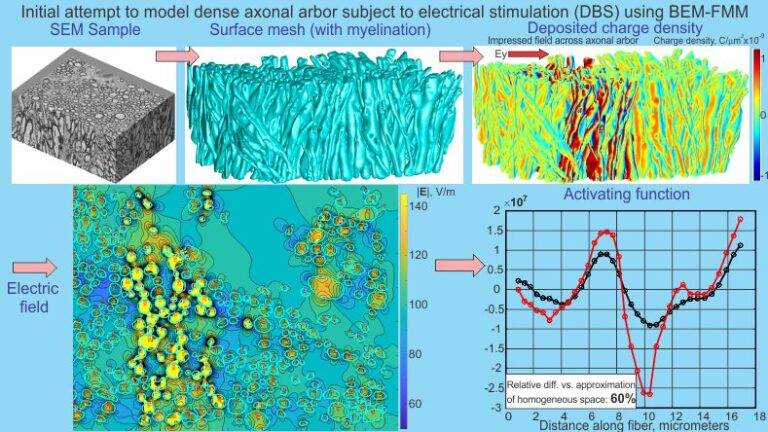
Biophysical models of neural stimulation are a valuable approach to explaining the mechanisms of neuronal recruitment via applied extracellular electric fields. Typically, the applied electric field is estimated via a macroscopic finite element method solution and then applied to cable models as an extracellular voltage source. However, the field resolution in this scenario is limited by the finite element size, typically 10’s-100’s of times greater than the average neuronal cross-section. As a result, induced charges deposited onto anatomically realistic curved membrane interfaces are not taken into consideration even though these details may significantly alter estimates of the applied electric field and predictions of neural tissue activation.
To characterize microscopic variations of the electric field, data for intra-axonal space segmented from 3D scanning electron microscopy of the mouse brain genu of corpus callosum were used. The boundary element fast multipole method was applied to accurately compute the extracellular solution. Neuronal recruitment was then estimated via an activating function.
Our results show that taking the physical structure of the arbor into account generally predicts higher values of the activating function. The relative integral 2-norm difference is 90% on average when the entire axonal arbor is present. A large fraction of this difference might be due to the axonal body itself. When an isolated physical axon is considered with all other axons removed, the relative integral 2-norm difference between the single-axon solution and the complete solution is 25% on average. Our result may provide an explanation as to why Deep Brain Stimulation experiments typically predict lower activation thresholds than commonly used FEM/Cable model approaches to predicting neuronal responses to extracellular electrical stimulation. The techniques provided herein may change methods for multiscale and bi-domain neural modeling and neural excitation, which warrant further exploration.