Yinchen Song, Rafael A. Torres, Sarahy Garcia, Yisel Frometa, Jihye Bae, Abhay Deshmukh Wei-Chiang Lin, Ying Zheng, and Jorge J. Riera, Florida International University, USA & University of Reading, UK
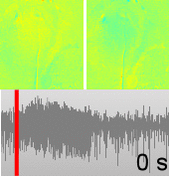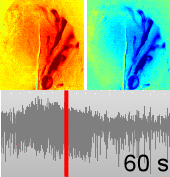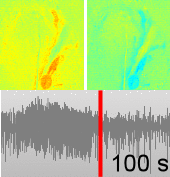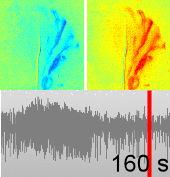
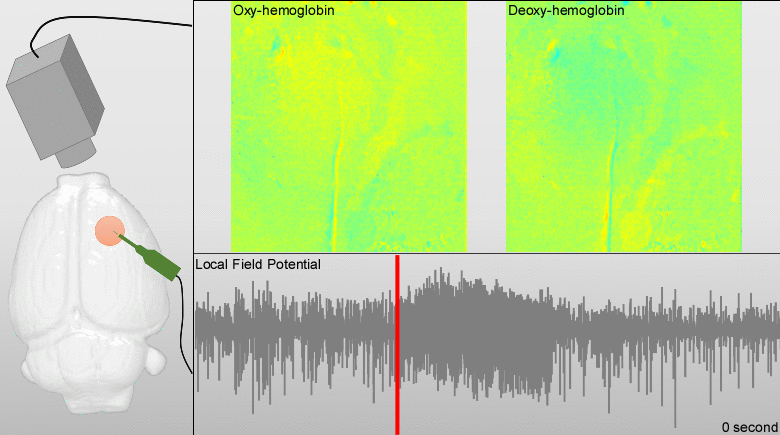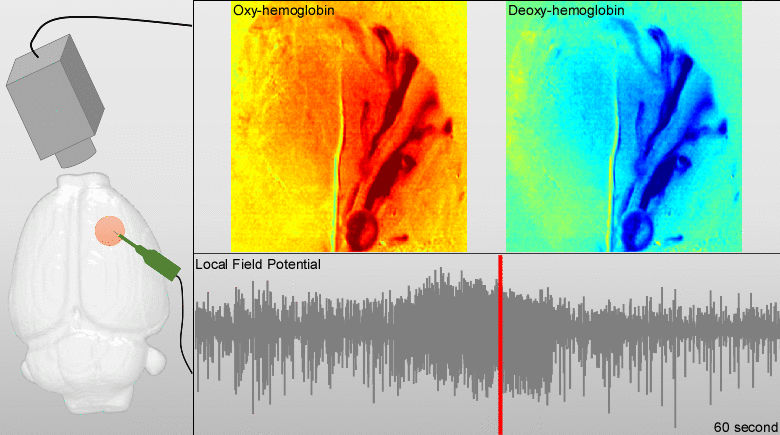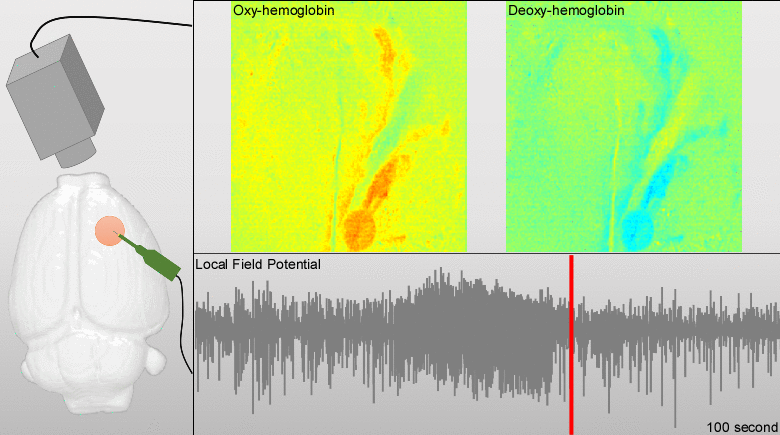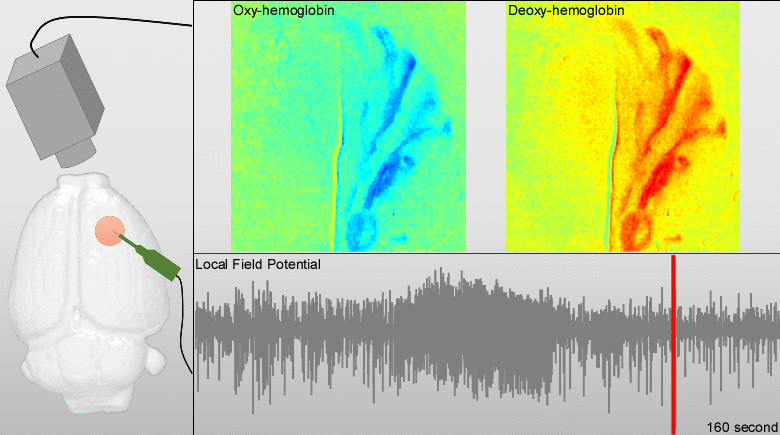
Dysfunctions in the neurovascular/metabolic coupling (NVMC) associated to epilepsy have been investigated using acute protocols for seizure genesis. However, no previous study has been performed to verify whether similar dysfunctions occur in a chronic epileptic cortex, which could have a tremendous impact on the interpretation and clinical use of neuroimaging data. In this article, we aim to evaluate mechanisms underlying the NVMC in the epileptogenic cortices of rats with chronic focal seizures. We performed and analyzed intracranial recordings obtained from the seizure onset zones of chronic epileptic rats during ictal periods, and then, used these data to fit a metabolically coupled balloon model. Normal rats undergoing forepaw stimulation with duration equivalent to that of seizures were used as control. Parameters linking the cerebral blood flow (CBF) and the relative concentration of deoxyhemoglobin to the neuronal activity in this biophysical model were significantly different between the epileptic and normal rats studied. The coefficient associated with the strength of the functional hyperemic response was significantly larger in the epileptogenic cortices, and changes in hemoglobin concentration associated with ictal activity reflected the existence of a significantly higher baseline for oxygen metabolism. In addition, we found a significantly higher contribution from the high frequency bands of local field potential to the CBF responses in the epileptogenic cortices during ictal neuronal activities. The hemodynamic responses associated with ictal activities were distance-dependent with regard to the seizure epicenter, though varied in profiles from those obtained from acute seizure models. The methods introduced in this article would enhance our understanding of the NVMC in epileptic brains and could have positive impact on the localization accuracy of the seizure-onset zones. This is also the first study capturing the spatiotemporal electrophysiological and hemodynamic signatures from in vivo brains of rats with chronic focal epilepsy during the ictal periods.
Keywords: chronic seizures, neuro vascular/metabolic coupling, epilepsy, biophysical models