Earlier this year, it appeared that the Ebola virus outbreak would be contained in West Africa; however, as seen of late, epidemics tend to be unpredictable. Instead, the Ebola virus has become an increasing concern and even more challenging since the first reported case from Guinea in March 2014. As of November 23, the World Health Organization reported at least 15,935 cases and 5,689 deaths in seven affected countries [1]. Given these statistics, the questions now are how far will it spread and at what rate? To mediate this situation, can a diagnostic test be developed to determine whether a patient is infected (exposed) but presents as asymptomatic in order to shorten or avoid quarantine? Overall, as epidemics continue to come and go, can we develop testing to differentiate between bacterial and viral infections? And as the global community embraces the Ebola virus epidemic, how will we move forward in the absence of effective early diagnostic tests and vaccines or therapeutics? Or is there a companion diagnostic test to monitor the effectiveness of a vaccine or drug in development?
The Ebola virus is a Filovirus that is comprised of five subspecies, four of which have been shown to cause infection in humans and of these four, three have high mortality rates. Patients infected with the Ebola virus present early signs and symptoms similar to that of many diseases making early diagnosis difficult. Such symptoms include sudden onset of fever, fatigue, muscle pain, headache, and sore throat, followed by vomiting, diarrhea, impaired kidney and liver function, and in some cases, both unexplained internal and external bleeding. Government officials are taking this matter seriously by implementing a 21-day quarantine for those infected (and/or those who have a history of traveling from West Africa) and those that came in contact with an infected individual, in order to break the transmission cycle [2]. Consequently, there is an immediate urgency to accelerate the development of vaccines and therapeutics to prevent and treat the disease and furthermore, diagnostic tools are needed to detect early onset of the disease because the initial signs and symptoms are so general, as well as a tool to measure the effectiveness of the therapeutic.
Ebola tests that are currently used or in development can only diagnose infection when the virus has replicated to levels high enough to be directly detected, typically 8-21 days after infection. At that stage, a person may already be contagious. Limitations in accurate diagnosis are either lengthy and/or often unnecessary quarantines. Unlike other viral infections, Ebola has evolved to evade detection by infecting dendritic cells (DCs) and inhibiting signaling of cytokines to eliminate the virus (avoiding the immediate antiviral effects of interferons (IFNs)) by Ebola viral protein 24 (eVP24) and eVP35, thus allowing the progeny of viral particles to invade cells, unchallenged, causing the infected cell to die and hemorrhage [3]. However, sub-types of IFNs (innate immune response) have shown to be expressed immediately after cells are infected with the virus, prior to the detection of elevated immunoglobulins, IgG and IgM (adaptive immune response), and the RNA of the virus. DCs are specialized cells that play a central role between the innate and adaptive immune responses. Upon infection, DCs have the ability to uptake particles and microbes by phagocytosis, process and then present them to other specialized immune cells, termed CD4+ T cells, which play a cardinal feature in the adaptive immune response. DCs are professional antigen presenting cells (APCs) that initiate the adaptive immune response by activating CD4+ T cells through at least three signals: 1) TCR activation, 2) activation of co-stimulatory molecules, and 3) secretion of chemokines and pro-inflammatory cytokines.
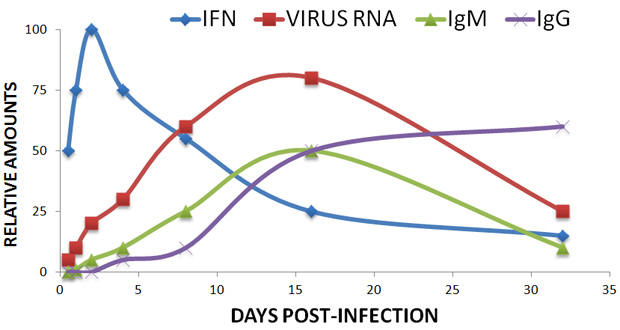
More importantly, these professional APCs can guide immune responses by inducing a specific CD4+ T cell polarization that is characterized by a particular cytokine release pattern, resulting in a specific immune response that targets specific pathogens [4]. Type I and II IFNs are among the tools utilized by DCs to regulate adaptive immune response and CD4+ T cell polarization. IFNs, a family of cytokines, constitute a first line of innate defense against viral infections; more specifically, type I IFNs, including IFNα and IFNβ, activating both toll-like receptor (TLR) and non-TLR signaling cascades [5]. This further underscores that the immunopathogenic properties underlying the Ebola virus are multifactorial and complex.
As many biotechnology and pharmaceutical companies are in the process of developing targeted therapeutics against the eVPs—where inhibiting the target would result in the increased production of type I IFNs—a potential prognostic biomarker for survival of patients infected with Ebola virus is possible. A co-diagnostic test with Ebola virus signatures and type I interferons would enable clinicians to monitor and/or measure the effectiveness of these experimental therapies until approved by the US Federal Drug Administration (FDA). This co-diagnostic test could also be utilized for screening where the IFN levels with patients infected would tend be low as compared to other viral infections like influenza and malaria, complementing existing Ebola tests.
We must prepare ourselves now and for the future as well as learn from prior epidemic experiences that include severe acute respiratory syndrome (SARS), H5N1 and H1N1 influenza, and the recent Middle East respiratory syndrome coronavirus (MERS-CoV), as there will be more unanticipated epidemics and outbreaks of new pathogens and/or re-emerging infections. In response, type I IFNs may play a critical role in not only the immune response, but mechanisms that interfere with the production of type I INFs are also potential targets for therapeutic development.
For More Information
References
- CDC: 2014 Ebola Outbreak in West Africa
- CDC: Ebola Signs and Symptoms
- Basler CF, Amarasinghe GK., Evasion of interferon responses by Ebola and Marburg viruses., J Interferon Cytokine Res. 2009 Sep;29(9):511-20. doi: 10.1089/jir.2009.0076.
- Kambayashi T, Laufer TM. Atypical MHC class II-expressing antigen-presenting cells: can anything replace a dendritic cell?, Nat Rev Immunol. 2014 Nov;14(11):719-30. doi: 10.1038/nri3754. Epub 2014 Oct 17.
- Hillyer P1, Raviv N, Gold DM, Dougherty D, Liu J, Johnson TR, Graham BS, Rabin RL., Subtypes of type I IFN differentially enhance cytokine expression by suboptimally stimulated CD4(+) T cells., Eur J Immunol. 2013 Dec;43(12):3197-208. doi: 10.1002/eji.201243288. Epub 2013 Oct 9.