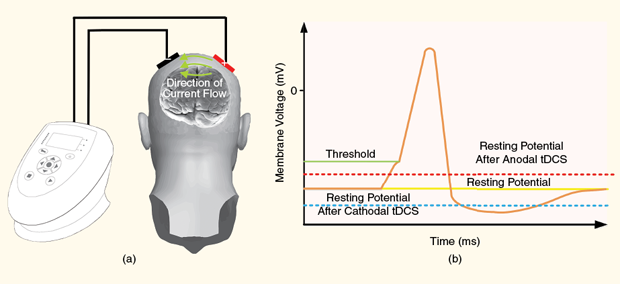
A treatment for depression, chronic pain, cravings, and more—this is the emerging field of transcranial direct current stimulation (tDCS). In tDCS, transcranial means through the skull, and direct current is a weak stimulation current delivered to the brain using noninvasive electrodes placed on the scalp (below, (a)).
Leading into the 2000s, the initial work on what is today called tDCS began in earnest [1]. Compared with other major neuromodulation techniques (such as transcranial magnetic stimulation and deep brain stimulation), tDCS boasts some key advantages: safety, ease of use, and relatively low cost. To date, tDCS has been applied to thousands of patients without causing serious adverse effects. There is good early evidence for applying tDCS to treat many conditions, including nicotine cravings, cognitive deficit, depression, poststroke rehabilitation, and pain (including both chronic and acute pain) [2]. These are conditions typically treated with drugs or surgery. There is also evidence that healthy people can improve cognitive functioning by receiving tDCS [2]. Beyond its applications as a clinical neuromodulation tool, tDCS has the potential to provide a new perspective on the mechanisms behind human perception, cognition, and action.
The main disadvantages of tDCS are that not all patients respond to treatment, dosage is not yet understood, long-term effects are unclear, and very large studies have yet to be carried out. The side effects of tDCS include mild tingling and itching sensations, fatigue, headache, and skin lesions at the site of electrode placement.
How and when tDCS works is still under investigation. In this article, we look at what goes into making and using a safe, state-of-the-art tDCS device and touch on the current challenges and biomedical engineering research.
tDCS Modulates Neural Activity by Enhancing or Depressing Firing Rates
The underlying mechanism behind tDCS may involve modifying how neurons communicate, affecting both the electrical activity of the neuron and the signal transduction process between neurons. In the neuron’s resting state, the cell maintains a negative electric potential across the cell membrane. When the potential of the cell membrane increases over the firing threshold, neurons generate (fire) an all-or-none action potential that propagates along the nerve fiber toward other neurons. By delivering current into the brain through electrodes applied to specific locations on the scalp [above, (a)], tDCS is thought to alter the resting potential [above, (b)] and, therefore, the likelihood that the neurons will fire. A positive flow of direct current is thought to increase the resting potential boosting neural excitation; conversely, negative current depresses firing rates.
In this way, tDCS can excite (anodal stimulation) or inhibit (cathodal stimulation) processes in the brain associated with specific brain regions. If the stimulation is of sufficient length, these effects are noticeable after the stimulation session is over. tDCS is thought to change neural connection strength in a manner analogous to long-term potentiation or long-term depression, meaning that increased firing is more likely to strengthen connections while a decrease in neural activation results in weaker connections.
Dose in the Context of tDCS
A tDCS dose can be defined as a series of parameters controlled by the practitioner that affect the interaction between the applied electric field and the biological tissue [3]. The factors that determine how the field is distributed spatially and temporally within the neural tissues, such as stimulation frequency, current intensity, and electrode size and position, are all part of the tDCS dose.
The selection of dose parameters should be based on maximizing treatment efficacy for a specific condition within an acceptable safety and risk profile. Clinical studies typically tend to use electrode sizes between 16 and 35 cm2, with current intensities set between 1 and 2 mA, usually administered for up to 20 minutes in duration, a period of time that has been empirically observed to induce post-treatment changes in the brain. Results of these studies show that current density as well as stimulation duration and frequency are strongly correlated with tDCS outcome, although quantifying and standardizing these effects have proven difficult.
The current density between the electrode and scalp is approximated as the quotient of current amplitude to electrode surface contact area. Therefore, the selection of electrode size is a critical part of determining tDCS dose: reducing the size of the electrodes may increase the current density maxima at the price of lower tolerance to stimulation. Based on preclinical studies on animal models, we know that current density levels must be at least two orders of magnitude higher than commonly used in tDCS to cause damage at a neural tissue level.
In summary, commonly accepted dose parameters are heuristically derived values based more on experience from initial clinical studies and the patient’s tolerability of the stimulation of the scalp rather than absolute safety thresholds required to prevent brain damage.
What Goes into Making and Using a Safe State-of-the-Art tDCS Device?
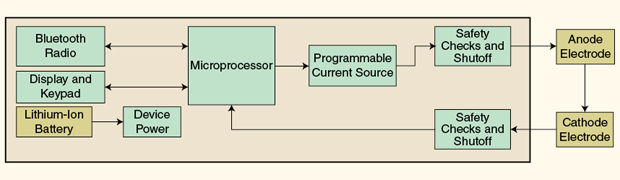
A high-level diagram (above) shows a functional overview of a modern tDCS device. A microprocessor controls a programmable current source, display, and keypad; communicates with a computer through a Bluetooth radio; and monitors the electrode connection quality. A programmable current source is used to ensure that current output remains constant in the event of tissue conductivity changes. It is known that the resistance of the scalp tissue may be highly variable over the course of the session. A safety system continuously monitors and compares current output and return to ensure consistent current intensity for the duration of the stimulation.
To avoid the patient experiencing a painful sensation with sudden changes in current intensity, stimulation usually starts by gradually increasing the current until the desired level and ends by gradually decreasing the current. Changing the current level occurs commonly over a 30-second ramp.
Before starting stimulation, it is important to ensure low electrode–tissue interface impedance, since poor electrode contact can result in painful or ineffective treatment. The quality of the electrode contact is dependent on the way the electrode is secured to the scalp, the amount and type of applied gel/saline, and local hair and scalp conditions. Connection quality at the electrode–tissue interface can be monitored by analyzing the overall impedance between the electrodes with short, low-intensity test pulses that are significantly below the treatment threshold.
tDCS has a control condition called “sham” stimulation that is used only for research purposes to control for the placebo effect. For the untrained trial subject, it is difficult to detect the difference between active and sham stimulation. tDCS devices used in clinical research generally can be programmed wirelessly for use in double-blind mode, where both subjects and researchers are unaware whether active or sham treatment is delivered.
Electrode materials—generally conductive rubber inserted into saline-soaked sponges—are used to ensure a homogeneous distribution of current over the stimulation target. The quantity of saline solution applied to the sponge should be chosen with care: excessive hydration may shunt the stimulation current between the sponges or beyond the electrode edges, while nonuniformly saturated sponges may cause high current density “hot spots.”
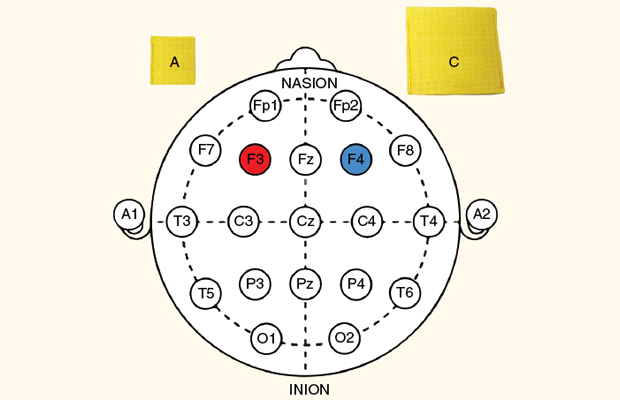
Accurate placement of the electrodes at the treatment site is a critical component for treatment success. The electrode placement scheme is called a “montage” and is defined by the International 10–20 system. Clinically, a montage is developed for a given condition based upon peer-reviewed clinical studies. The figure below shows a left dorsolateral prefrontal cortex anodal tDCS montage, which represents one potential montage for depression. The cathode can be much larger than the anode.
Challenges and Biomedical Engineering Research Directions
The proportion of the electrical dose reaching the brain area of interest is very difficult to determine and is greatly influenced by the electrical properties of the layers that enclose the brain. As the scalp is much more conductive than the skull, much of the current is shunted from one electrode to the other through the scalp, never reaching the brain [as shown in above, (a)]. The current that does penetrate through the skull is further dissipated through the conductive cerebrospinal fluid. Shunting prevents focal and accurate treatment.
Even if the current dose reaching the desired brain region was known, the effect of the stimulation field on neural processes such as plasticity and modulation are far from being understood. Given these major obstacles to controlling tDCS, any simple model of tDCS as electrical “brain boosting” is inaccurate and misleading. It is hardly surprising that some clinical subjects do not respond to tDCS treatment. Even some of those who do respond to treatment may have varying outcomes from one treatment to the next.
Strategies to reduce the variability of tDCS outcome include both new electrode design to improve focality and use of models to better understand and estimate the current density. Several researchers have developed advanced finite-element head models to accurately simulate the current propagation path into the brain. Computational modeling is increasingly being used to support experimental approaches to improve current focality, for example, by decreasing the size of the electrodes. Other approaches include the introduction of electrode arrays and shaping current by controlling input current of the electrode arrays.
To further understand the relationship between current dose and outcome, it is important to have reliable methods of obtaining feedback indicative of physiological response. Examples include monitoring electroencephalogram (EEG) signals during stimulation and measuring variation in blood oxygenation levels in cortical tissue using functional near-infrared spectroscopy and functional magnetic resonance imaging.
A goal of future research is to utilize physiological feedback to directly quantify individual dose response to stimulation parameters both in real time as well as in offline analysis. Other research directions include developing methods to increase the standardization and reproducibility of clinical trials. This may involve obtaining real-time information, such as the voltage between the electrodes, pressure applied to the electrode, scalp impedance, or other data that might be useful for obtaining individual specific treatment data that could be correlated to physiological response.
These research challenges will require significant involvement of biomedical engineers and researchers for years to come.
Transitioning from the Research to Mainstream Clinical Use: Will It Happen Soon?
The rate of tDCS publications have been steadily increasing throughout the last decade. All indications are that this is not going to slow down anytime soon as the popularity of this intuitively simple yet ultimately complex technology spreads through research labs around the world. This heightened research activity is mirrored by increased interest of nonacademic groups like popular forums and the do-it-yourself community. The perception of tDCS as a speculative research tool is slowly changing, as evidenced by the increased flow of capital to commercialize this technology.
Despite these positive signs, there is cause for some concern, and there is still much that is unknown. The efficacy of tDCS, especially in terms of long-term effects, is increasingly being challenged. Regulators and insurance providers have taken a cautious wait-and-see approach until efficacy can be clearly demonstrated by large, controlled clinical trials. Attempts to introduce tDCS as a mainstream therapeutic offering appear to be gaining momentum in select areas, such as chronic pain, but only time will tell if this surge of interest is of a temporary or targeted nature or an indication of a breakthrough transformative clinical therapy with myriad applications.
Disclaimer
Herschel Caytak, Daniel Shapiro, and Alexey Borisenko hold shares or stock options in Nuraleve Inc., a tDCS company serving the chronic pain and cravings markets. Miodrag Bolic has received research funding from Nuraleve. Alexey Borisenko is employed by Nuraleve, while Daniel Shapiro was employed by Nuraleve.
Acknowledgments
We would like to thank Mitacs, the National Research Council Canada–Industrial Research Assistance Program (NRC-IRAP), Ontario Centres of Excellence, Connect Canada, the Natural Sciences and Engineering Research Council of Canada, and Nuraleve Inc. for their financial support.
References
- M. A. Nitsche and W. Paulus, “Excitability changes induced in the human motor cortex by weak transcranial direct current stimulation,” J. Physiol., vol. 527, no. 3, pp. 633–639, Sept. 2000.
- R. C. Kadosh, The Stimulated Brain: Cognitive Enhancement Using Non-invasive Brain Stimulation. Amsterdam, The Netherlands: Elsevier, 2014.
- A. V. Peterchev, T. M. Wagner, P. C. Miranda, M. A. Nitsche, W. Paulus, S. G. Lisanby, A. Pascual-Leone, and M. Bikson, “Fundamentals of transcranial electric and magnetic stimulation dose: Definition, selection, and reporting practices,” Brain Stimulation, vol. 5, no. 4, pp. 435–453, Oct. 2012



