FROM EYE DISEASE TO NEUROLOGICAL CONDITIONS, NEW TECHNOLOGIES ARE MAKING EARLIER DIAGNOSIS AND BETTER PATIENT OUTCOMES A REALITY BY INVESTIGATING THE EYE.
The eyes are said to be the windows of the soul, but they are increasingly the door to diagnostic information that could transform the lives of millions of people. Age-related macular degeneration (AMD), diabetic retinopathy, and glaucoma are the big three causes of permanent loss of vision, while severe neurological diseases like Alzheimer’s and Parkinson’s are on the rise. Thanks to new technologies for imaging the eye, the early signs of all of them can be detected earlier.
According to the National Eye Institute (NEI), the number of people in the USA with glaucoma is expected to more than double, from 2.7 to 6.3 million by 2050. Over the same period, the number of people with AMD is expected grow at a similar rate from 2.07 to 5.44 million. Technological innovation could, however, have a major impact on these trends.
“Humans might have seen the world for centuries, but the world has only been able to see inside the living human eye for the last 170 years,” says Prof. Parsin Haji Reza of the University of Waterloo, Waterloo, ON, Canada, who develops novel solutions for clinical and pre-clinical biomedical applications. “The eye gives us the most important sense of a human being and I believe it needs a lot of attention in research and development.”
An auspicious accident
Current state of the art methods, such as fundus imaging and optical coherence tomography (OCT), which are the primary diagnostic tools used in clinic, focus on measuring morphological information about the structures in the eye, capturing factors like layer integrity and shape, or blood vessel diameters. “Right now, there is no commonly available system that can measure key functional parameters in the retina, including blood oxygenation, or metabolic rate of oxygen consumption, which more directly measure the health of the eye,” says Haji Reza.
Since 2018, Haji Reza has been developing photoacoustic remote sensing (PARS) microscopy, the first ophthalmic imaging tool capable of pre-diagnosing eye-blinding diseases before they become symptomatic. “The idea for PARS came accidentally during a different experiment, when I saw the possibility of detecting photoacoustic signals remotely,” he adds. “I was not considering academia but when I discovered PARS during my Ph.D., I realized that this technology, like any other infant technology, needs proper focus and mindset to become mature.”
PARS differs from traditional photoacoustic imaging systems, which use a pulsed laser to excite a specific biological target, and an ultrasound transducer to measure the generated acoustic wave. While these methods have led to great strides forward in the imaging of biological tissues, the need for the ultrasound transducer to be in contact with the sample being imaged means that applications in key fields such as ophthalmology were limited. PARS overcomes this limitation by measuring the changes in intensity of a co-focused continuous-wave laser to detect the photoacoustic signal (Figure 1). It brings all the same benefits but requires no physical contact.
“This enables us to move into key application spaces including functional imaging of the retina or in-situ imaging of tissues in surgery,” says Haji Reza. “PARS has shown better penetration depth compared to high-resolution photoacoustic methods. It also offers the ability to directly measure absorption contrast, and as a result can capture these key functional parameters to better characterize eye tissue health.”

With this kind of data gathered regularly, clinicians could observe decreases in tissue health before they show up in the morphological characteristics of the eye. “Seeing the problem earlier means we can also take preventative steps earlier to stop progression before it has a chance to impact the delicate structures in the eye,” he adds. “Right now, we don’t know how much earlier PARS retina imaging could potentially detect these serious conditions, but it is well known that functional information better represents key diagnostic markers including tissue health which should help push that diagnosis date earlier in time.”
OCTA opens the door to the brain
Since it was first conceived by Professor Jim Fujimoto from the Massachusetts Institute of Technology (MIT), Cambridge, MA, USA, back in 1991, OCT has developed step-by-step to become one of the most widely used technologies for assessing eye health. The technology can quantify blood flow in larger vessels in the eye and measure total retinal blood flow, both key indicators of eye disease.
Over the 30-year development, Fujimoto has worked closely with Professor David Huang from Oregon Health & Science University, Portland, OR, USA, who handles algorithm development to explore specific clinical applications, and Jay Wei, who recently retired from Optovue, the company that developed the first spectral domain OCT instrument in the USA. The data provided by OCT goes beyond the 2D images, which only give a surface snapshot of the retina. It provides data on the many deeper layers of eye tissue, showing the depth of specific structures and measuring how much it reflects or scatters light (Figure 2). This was revolutionary, but since 2010 it has been the standard technology for retinal analysis.
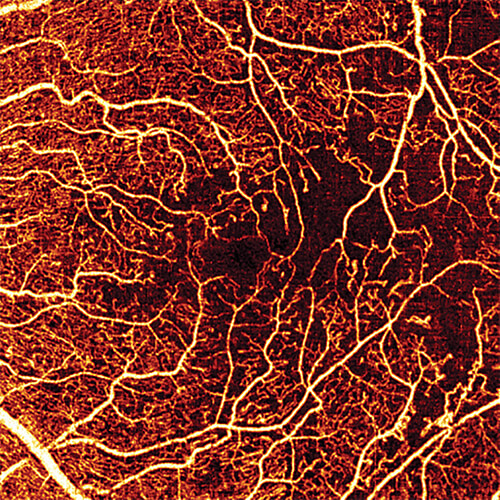
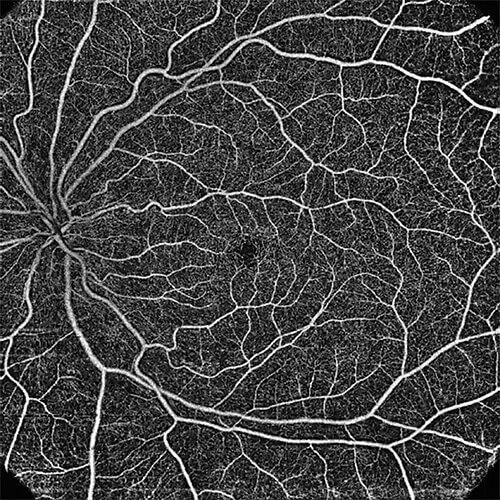
Fast forward to 2014, and the first clinical studies of OCT angiography (OCTA) appeared. OCTA provides unparalleled depth of information by adding the dimension of time to 3D OCT images. It can analyze blood flow with unprecedented resolution through all vascular layers of the retina in a rapid, noninvasive fashion, using repeated B-scans to detect motion contrast and visualize vasculature (Figure 3).
“It provides 3D images of how structures have changed over time,” Wei explains. “In diabetic retinopathy, for example, you can only see messy results from a photo. Too little blood flow causes problems and too much blood flow can cause weak tissue to break. With traditional fluorescein angiography, you need to inject dye, which is time-consuming and not a simple procedure. With OCTA, no dye is needed. You don’t even need to dilate the pupil and you can easily spot where there is a problem with blood flow.”
Though yet to become mainstream, OCTA already promises much earlier diagnosis of the big three threats for vision loss. But its resolution is so high and the detail of its images so precise that it is already finding new applications outside of ophthalmology. “My team is working on algorithms and signal processing to more easily identify low or non-profusion areas and to detect abnormal blood flow,” says Huang. “Neurological and neurovascular diseases are also being looked at with OCTA as they leave signatures in the eye.
The retina is a small piece of the central nervous system and faults in other parts of the body are reflected in the eye.”
The eye is so close to the brain that it can be the window through which to see the earliest sings of neurodegenerative diseases. OCTA’s ability to precisely monitor blood flow can reveal artifacts that were previously invisible.
“Early diagnosis is key,” says Srinivas Sadda from the Doheny Eye Institute, University of California, CA, USA, who uses OCTA extensively. “With Alzheimer’s, brain alterations come many years before dementia presents, but OCTA can detect attenuation of retinal vessels. Abnormalities seen using the technology can also reveal signs of Parkinson’s.”
OCTA is just beginning to reveal its potential, and work continues to not only improve its resolution, but also to harness the power of artificial intelligence (AI) to analyse the vast amount of data OCTA scans produce. Among so many images, it may be difficult to spot the key danger signs, but with AI flagging images that need further investigation by a clinician, the results could be better, faster, and more crucial in determining patient outcomes.

EyeQue puts eye health in the patient’s hands
Far from university laboratories and ophthalmology clinics, the latest innovation in eye health sits in the palm of a patient’s hand. Miami-based company EyeQue has developed a fast, affordable, and convenient way for patients to test their own eyes and, using results expressed as EyeGlass Numbers (EGNs), order new glasses from the comfort of their own home.
The VisionCheck device is an optical smartphone attachment that performs a refraction test rivaling traditional room-sized ophthalmic refractometers in terms of accuracy (Figure 4). “Since 2010, smartphone screens have had sufficiently high resolution that we can use the light from the screen to measure the refraction of the eye,” says John Serri, resident and the co-founder of EyeQue, who along with Tibor Laczay developed VisionCheck from a technology patented by MIT. “This is a disruptive technology because it is a low-cost device that takes something out of the domain of optometrists.”


In 2017, the company launched the device, which uses three lenses, some slits, and rotational elements to refract the light from the screen. Made entirely from plastic, it contains no electronics and is simple enough to operate that with minimal instruction patients can get an accurate reading of the quality of their vision (Figures 5 and 6).
“Will it replace optometrists? No,” says Serri. “It makes accurate measurements, but it does not examine eye health. What’s unique about it is that you can track the changes in your vision over time, and you can do as many tests as you want. In fact, the more you use it, the greater the accuracy the back-end software can deliver from your data.”
VisionCheck is big step toward telemedicine in the field of eye care. According to a recent survey from the COVID-19 Healthcare Coalition, almost 80% of people who used telehealth during COVID-19 reported patient satisfaction, and over 70% expect to access virtual care post-pandemic.
“EyeQue is entering telemedicine now,” says Serri. “Patients love it, and doctors love it. You can’t bring home a $20,000 phoropter from the optometrist’s clinic, but you can easily use VisionCheck.”
Delivering self-testing and telemedicine, as well as enabling detailed investigation of major diseases in the eye and brain, innovation in eye care is happening at an unprecedented rate. They eyes may soon become not only the windows to the soul, but the gateway to human health.



