Abstract
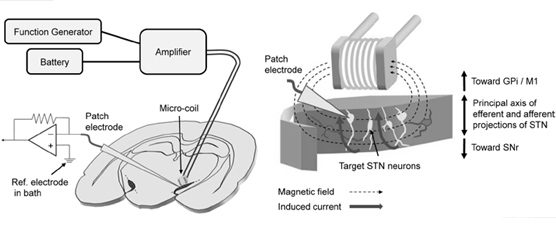
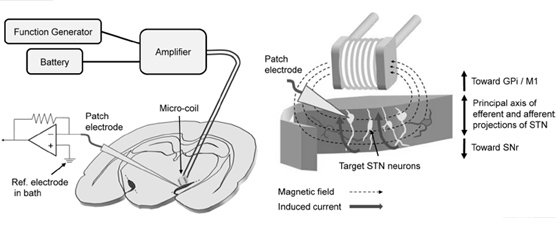
Magnetic stimulation delivered via 0.5-mm diameter coils was recently shown to activate retinal neurons; the small coil size raises the possibility that micromagnetic stimulation (μMS) could underlie a new generation of implanted neural prosthetics. Such an approach has several inherent advantages over conventional electric stimulation, including the potential for selective activation of neuronal targets as well as less susceptibility to inflammatory responses. The viability μMS of for some applications, e.g., deep brain stimulation (DBS), may require suppression (rather than creation) of neuronal activity, however, and therefore we explore here whether (μMS) could, in fact, suppress activity. While single pulses elicited weak and inconsistent spiking in neurons of the mouse subthalamic nucleus (in vitro), repetitive stimulation effectively suppressed activity in ~70% of targeted neurons. This is the same percentage suppressed by conventional electric stimulation; with both modalities, suppression occurred only after an initial increase in spiking. The latency to the onset of suppression was inversely correlated to the energy of the stimulus waveform: larger amplitudes and lower frequencies had the fastest onset of suppression. These findings continue to support the viability of μMS as a next-generation implantable neural prosthetic.