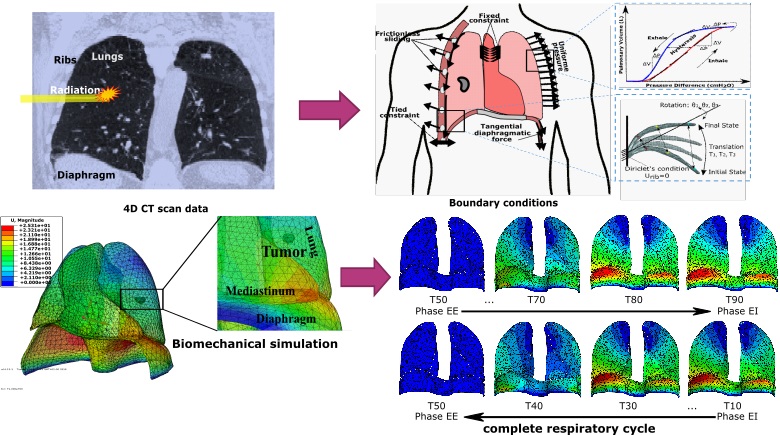
Respiratory-induced organ motion is a technical challenge to medical imaging, nuclear imaging, and radiation therapy for lung cancer treatment in particular. In this context, we present a new lung tumor tracking approach based on a patient-specific biomechanical model of the respiratory system, which takes into account the physiology of respiratory motion to simulate the real non-reproducible motion. The behavior of the lungs, is directly driven by the simulated actions of the breathing muscles, i.e. the diaphragm and the intercostal muscles (the rib cage). The lung model is monitored and controlled by a personalized lung pressure/volume relationship during a whole respiratory cycle. The lung pressure and rib kinematics are patient-specific and obtained by surrogate measurement. The ribs displacement is computed by finite helical axis transformation, from the end of exhalation (EE) to the end of inhalation (EI). The lung pressure is calculated by an optimization framework based on inverse finite element analysis, by minimizing the lung volume errors, between the respiratory volume (respiratory airflow exchange) and the simulated volume (calculated by biomechanical simulation).
We have evaluated the model accuracy on five public datasets. We have also evaluated the lung tumor motion identified in 4D CT scan images and compared it with the trajectory obtained by finite element simulation. The effects of rib kinematics on lung tumor trajectory have also been investigated. Over all phases of respiration, our developed model is able to predict the lung tumor motion with an average landmark error of 2.0 ± 1.3mm. The results demonstrate the effectiveness of our physics-based model.
We believe that this model could successfully be used as non-invasive lung tumor tracking system during beam therapy based on external sensors, in 4D dose deposit computation as well as breathing motion artifacts correction in positron emission tomography (PET) or gamma prompt image reconstruction.

