Single ventricle defects are among the most severe types of congenital heart defects. These patients undergo several palliative surgeries culminating in the Fontan circulation which routes the entire venous return directly to the lungs without going through a sub-pulmonary ventricle. A significant number of Fontan patients ultimately develop circulatory failure. Even though cardiac transplantation is considered throughout the patient’s lifetime, many patients are not able to receive transplantation as a result of multiple organ dysfunctions as well as unavailability of matching organ donors.
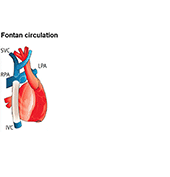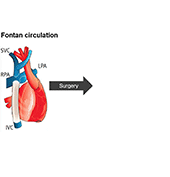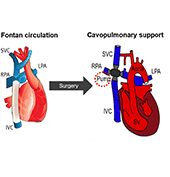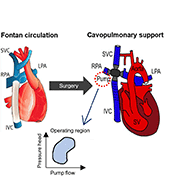
The use of a mechanical assist device to support the cavopulmonary segment of the circulation has been proposed to reenergize the returning venous return to the lungs. However, there is currently no device commercially available for the cavopulmonary support application and little research to inform the design criteria of such a device. A suitably designed cavopulmonary assist device should have performance profiles versatile enough to serve the range of failing Fontan patients with various levels of hemodynamic instability.
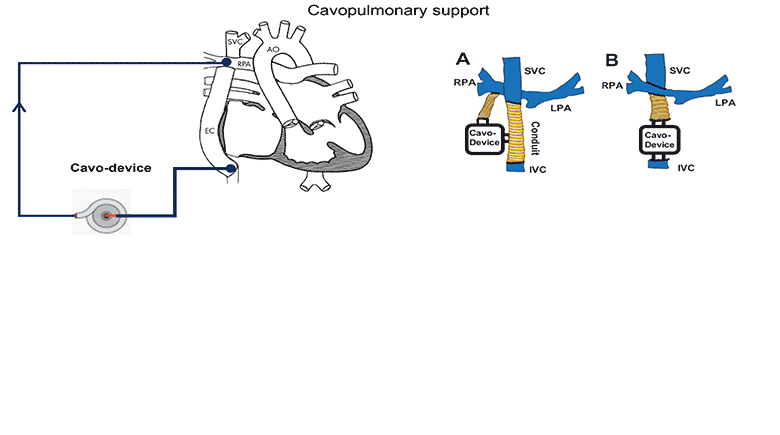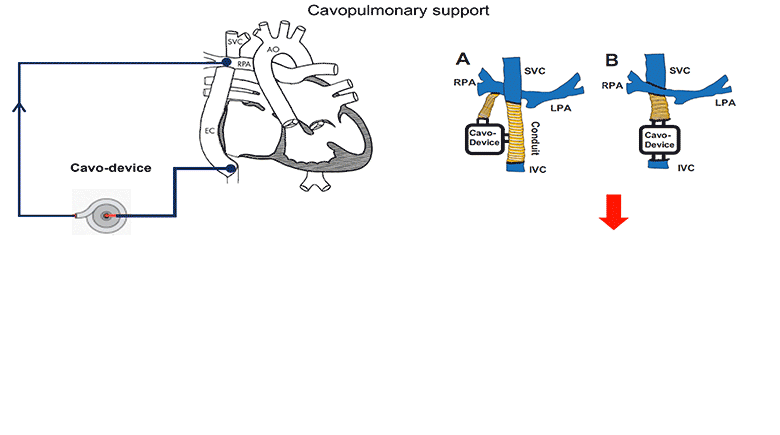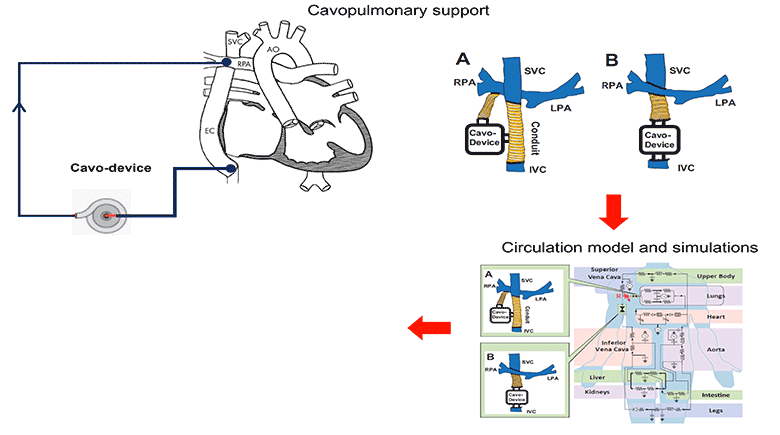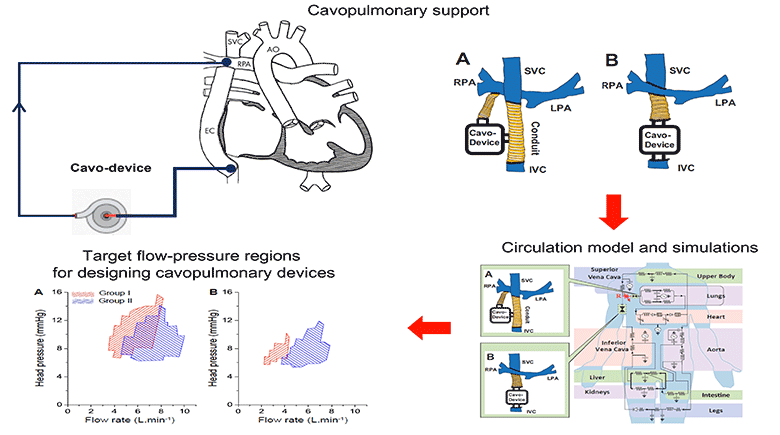
We simulated the interaction of a cavopulmonary assist device with a full range of failing stage Fontan physiologies, reflecting pathological conditions with various severity levels and encompassing approximately 95% of the adult failing Fontan physiologies. From the analysis of these simulation results we report the appropriate hydraulic operating regions for a failing Fontan cavopulmonary support device. These flow-pressure operating regions can also serve as the criteria for evaluating the off-label use of commercially available left-side blood pumps for failing Fontan cavopulmonary support. Furthermore, this study identifies physiologic scenarios where cavopulmonary support would not benefit patient hemodynamics due to the risks of perfusion lung damage or venous collapse as a result of the pressure generated by the cavopulmonary device. These results may help provide guidance for identifying patients unsuitable for cavopulmonary support.