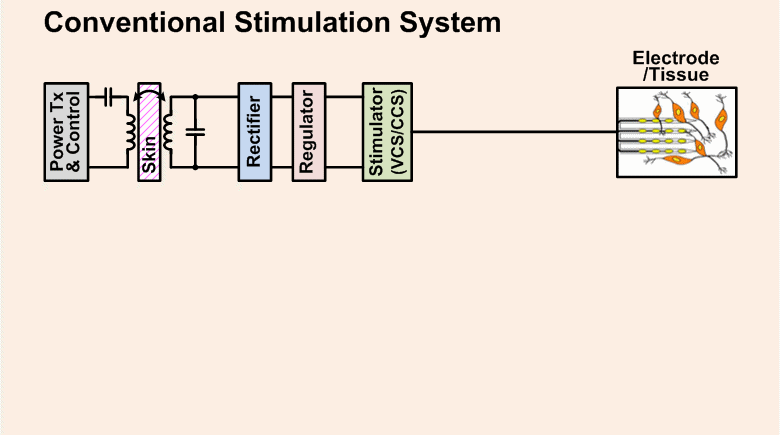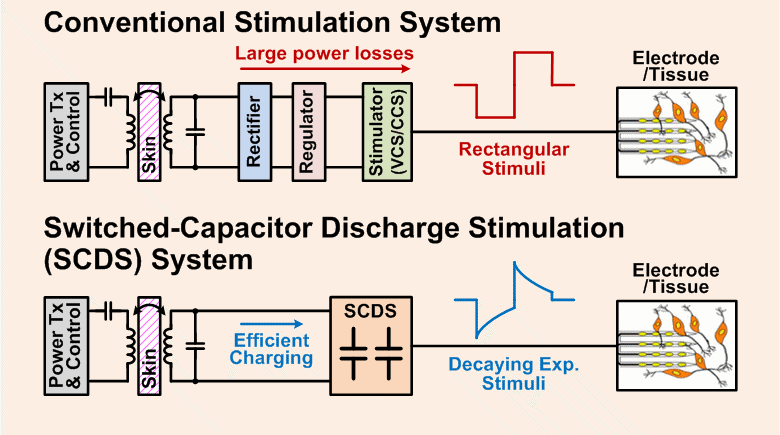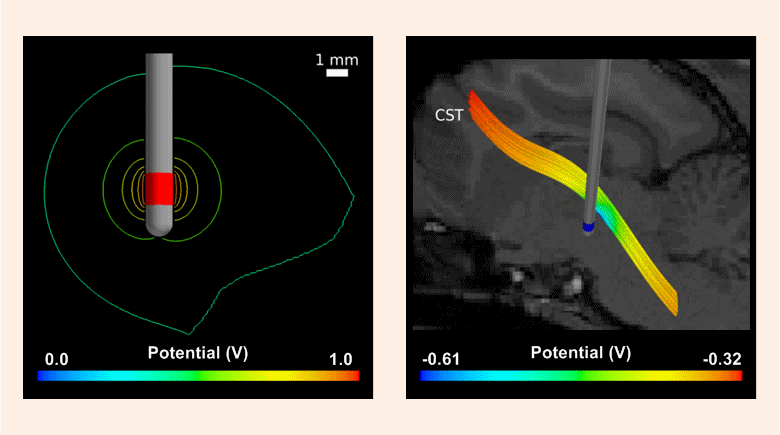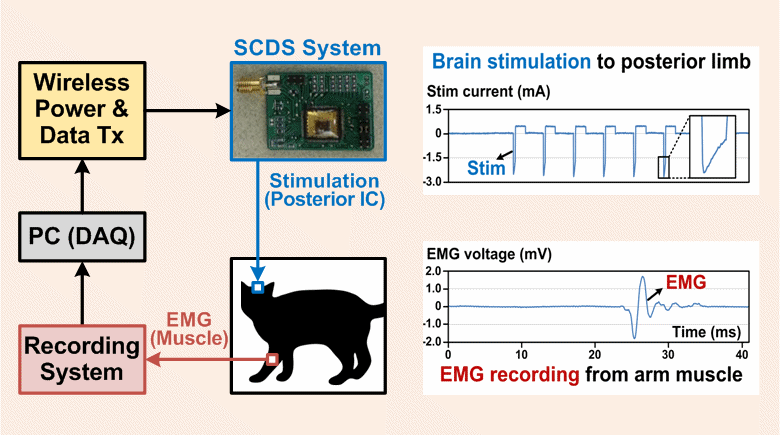
Implantable pulse generators (IPGs) that use a switched-capacitor discharge stimulation (SCDS) system and are wirelessly powered via an inductive transcutaneous link can be made significantly smaller than conventional IPGs powered by implantable batteries. The SCDS system is more energy-efficient at charge injection than conventional stimulation systems, but its efficiency at stimulating neural elements has not been quantified. The purpose of this study was to test the feasibility of using a switched-capacitor discharge stimulation (SCDS) system for electrical stimulation, and, subsequently, determine the overall energy saved compared to a conventional stimulator.
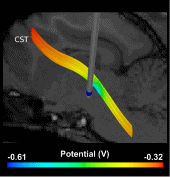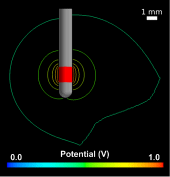
In this work, we have constructed a computational model by pairing an image-based volume conductor model of the cat head with cable models of corticospinal tract (CST) axons and quantified the theoretical stimulation efficiency of rectangular and decaying exponential waveforms, produced by conventional and SCDS systems, respectively. Subsequently, the model predictions were tested in vivo by activating axons in the posterior internal capsule and recording evoked electromyography (EMG) in the contralateral upper arm muscles.
Compared to rectangular waveforms, decaying exponential waveforms with time constants >500 μs were predicted to require 2%–4% less stimulus energy to activate directly models of CST axons and 0.4%–2% less stimulus energy to evoke EMG activity in vivo. Using the calculated wireless input energy of the stimulation system and the measured stimulus energies required to evoke EMG activity, we predict that an SCDS implantable pulse generator (IPG) will require 40% less input energy than a conventional IPG to activate target neural elements.
A wireless SCDS IPG that is more energy efficient than a conventional IPG will reduce the size of an implant, require that less wireless energy be transmitted through the skin, and extend the lifetime of the battery in the external power transmitter.
Research Websites: