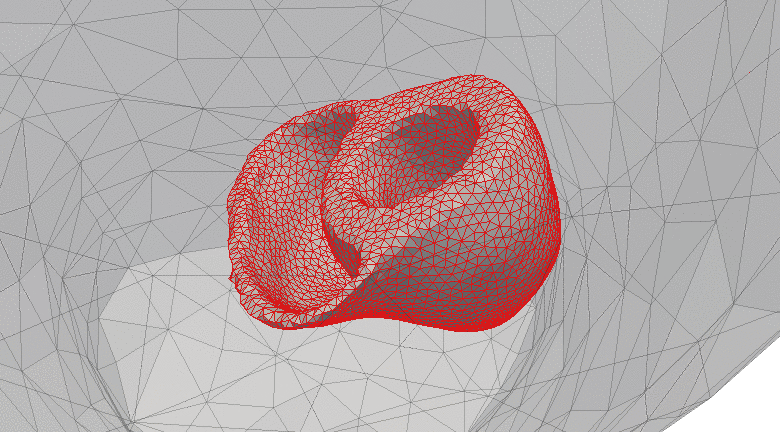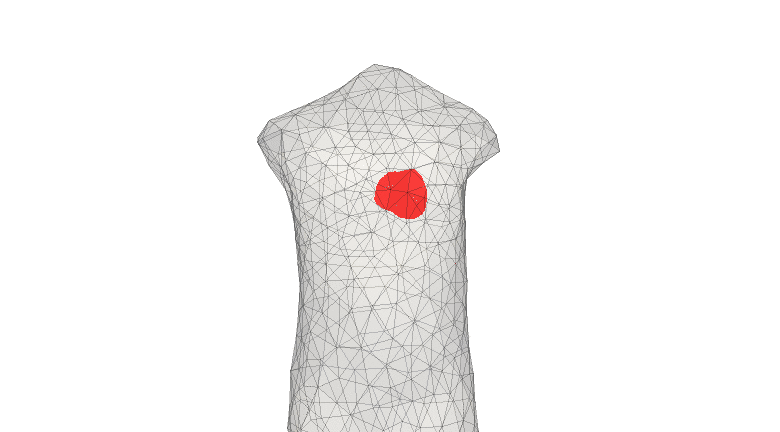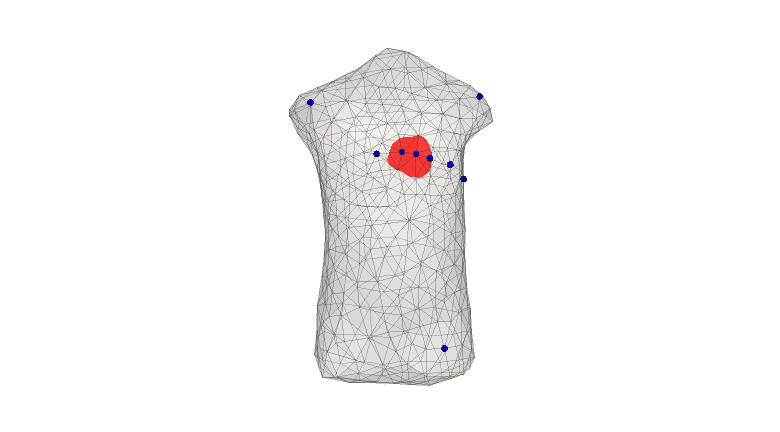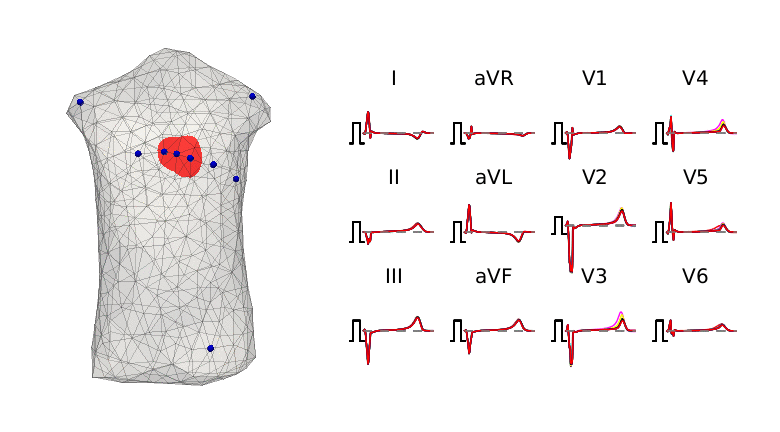
There is a pressing clinical need to identify developing heart attack (infarction) in patients as early as possible. However, current state-of-the-art tools in clinical practice, which are often underpinned by evaluation of elevation of the ST-segment of the 12-lead electrocardiogram, do not identify all patients suffering from lack of blood flow to the heart muscle (cardiac ischemia), worsening the risk for further adverse events and patient outcome overall. In this study, we aimed to explore and compare the portions of cardiac repolarization in the ECG that best capture the electrophysiological changes associated with ischemia via a fundamental engineering approach based on organ-level forward simulations of electrodynamics. We developed 3D electrophysiological models of the human ventricles and torso, incorporating biophysically-based membrane kinetics and realistic activation sequence. These models were used to compute simulated ECGs and to examine ECG alteration with application of simulated ischemia of differing severity in diverse regions of the simulated heart. The ischemic volumes were varied in size from diameters of 1cm up to 4cm; 17 different locations in the left ventricles were tested.
For each location (17), ischemic severity (4) and ischemic state (3), (in all 204 cases), we computed the resulting ECG and compared this to the healthy reference ECG in order to quantify the observability of the different cases. Our results suggest that metrics based on the portion of the electrocardiogram representing heart repolarization, the T-wave, are more sensitive to and better highlight the presence of developing ischemia than the metric of standard practice. Further research into how such simulation-aided risk assessment methods may aid workflows in extant clinical practice, with the ultimate goal of multi-modality clinical support, is warranted.

