Ibrahim A. Elabyad, Raffi Kalayciyan, Nagesh C. Shanbhag, and Lothar. R. Schad, Heidelberg University, Germany
Volume 61, Issue 2, Page: 334-345

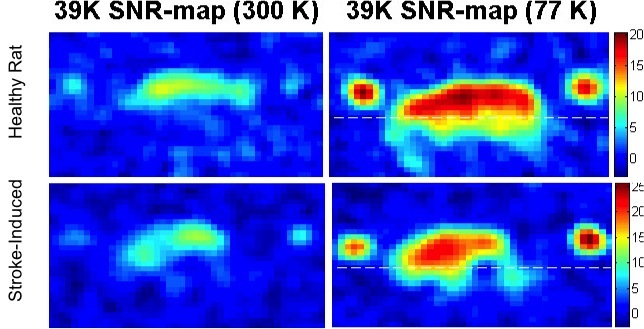
Potassium-39 (39K) magnetic resonance imaging (MRI) is a noninvasive technique which could potentially allow for detecting intracellular physiological variations in common human pathologies such as stroke and cancer. However, the low signal-to-noise ratio (SNR) achieved in 39K-MR images hampered data acquisition with sufficiently high spatial and temporal resolution in animal models so far. Full wave electromagnetic (EM) simulations were performed for a single loop copper (Cu) radio frequency (RF) surface resonator with a diameter of 30 mm optimized for rat brain imaging at room temperature (RT) and at liquid nitrogen (LN2) with a temperature of 77 K. A novel cryogenic RF surface resonator with home-built LN2 nonmagnetic G10 fiberglass cryostat system for small animal scanner at 9.4 T was designed, built and tested in phantom and in in vivo MR measurements. Aerogel was used for thermal insulation in the developed LN2 cryostat. The 3D chemical shift imaging (CSI) sequence was used for 39K-MRI at 9.4 T since the short relaxation times T1 allow for very short repetition times (TRs). In this work, we present the first in vivo 39K-MR images at 9.4 T for both healthy and stroke-induced rats using the developed cryogenic coil at 77 K. In good agreement with EM-simulations and bench-top measurements, the developed cryogenic coil improved the SNR by factor of 2.7 ± 0.2 in both phantom and in in vivo MR imaging compared with the same coil at RT. Results show that 48 hours after middle cerebral artery occlusion (MCAO), the 39K signal in the occluded hemisphere of the rat brain has been decreased by about 40% at 300 K and 75% at 77 K compared to the non-occluded hemisphere. The significant variations in 39K signal after ischemic stroke were successfully measured non-invasively, which could help to understand the physiological variations after ischemic brain damage.