L. Alan Bradshaw, Juliana H. Kim, Suseela Somarajan, William O. Richards, and Leo K. Cheng, Vanderbilt University, Lipscomb University, University of South Alabama, USA, Auckland University, New Zealand
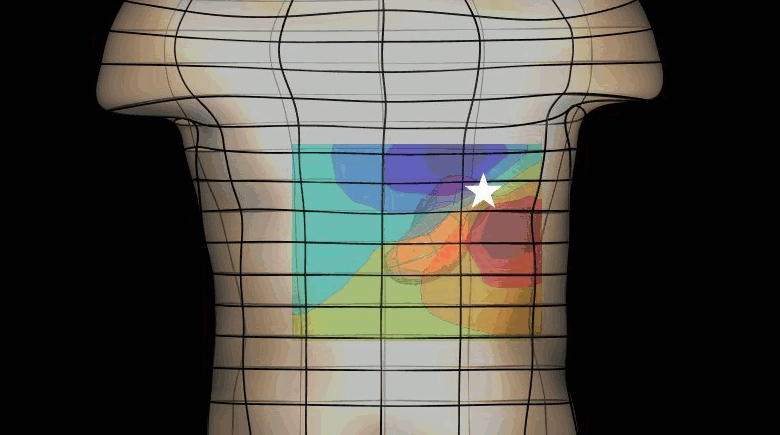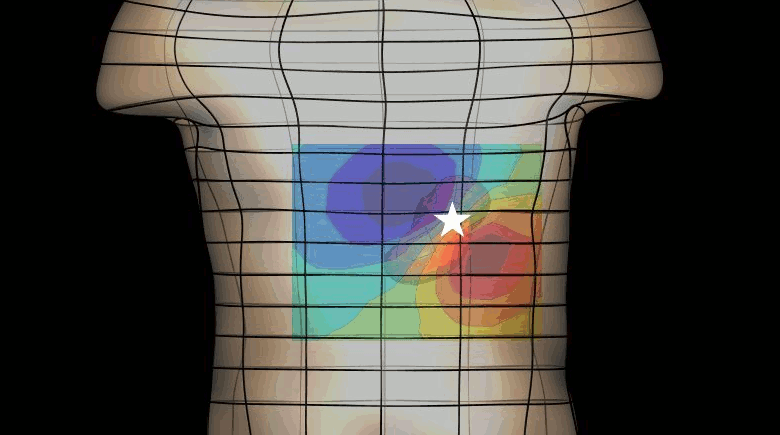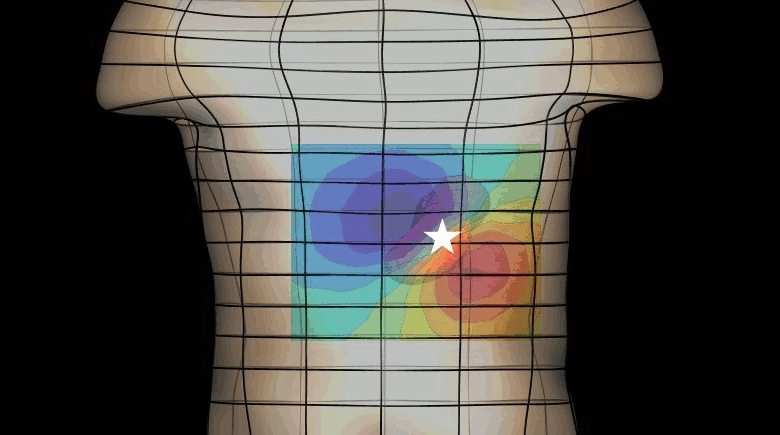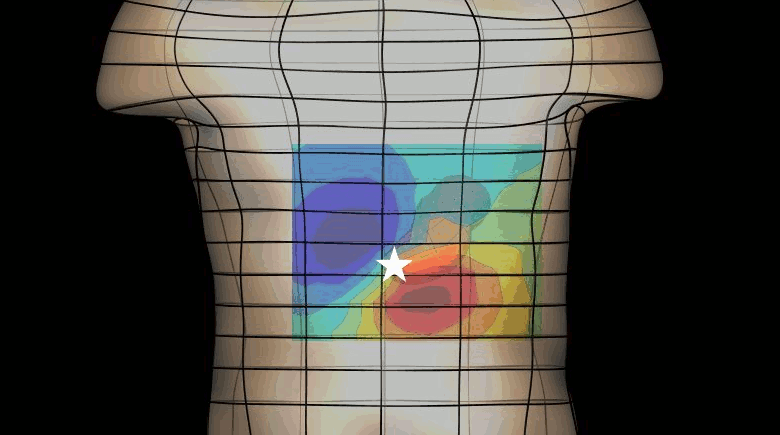
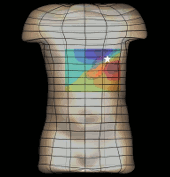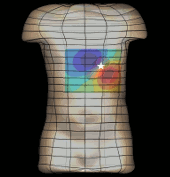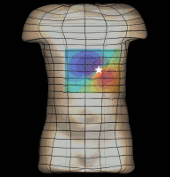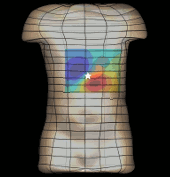
The magnetogastrogram (MGG) is one example of propagating activity from a bioelectric source that may be a clinically significant indicator of physiological function. We created a mathematical model of transabdominal magnetic fields (the MGG) from propagating gastric electrical slow waves. Propagating current dipoles were used to represent the gastric slow wave inside a realistic abdominal model and a horizontal half-layered volume conductor. We simulated the MGG recorded by a hypothetical 110-sensor Superconducting QUantum Interference Device (SQUID) biomagnetometer, an instrument with sensitivity sufficient to detect bioelectric currents. Two methods to analyze propagation were compared. Second Order Blind Identification (SOBI) is a blind source separation technique that identifies signal components in multichannel data and excludes noise contributions. The SOBI-reduced MGG spatiotemporal map contains gastric signal sources reconstructed from MGG and allows tracking of map features to analyze propagating sources. Surface Current Density (SCD) mapping uses differences between adjacent MGG channels to estimate of the current density at the surface of magnetometer, which hypothetically represents the internal source current.
Mean square errors in position estimates were within 2 cm of the correct source position and average propagation velocities within 2 mm/s of the actual slow wave source propagation velocity. SOBI propagation analysis proved more robust than the SCD method except for deep sources. SOBI analysis of propagation was also less sensitive to additive noise. These methods were further tested in porcine experiments, where the serosal electromyogram in a 48-channel electrode array was measured simultaneous with transabdominal MGG. Propagation velocities using both SOBI and SCD maps agreed with propagation velocity determined from the serosal EMG array. Although this model was developed specifically for gastric electrical activity, the methods for analysis of propagation are easily extended to multichannel recordings of any propagating activity.
Keywords: Bioelectric phenomena, SQUIDs, biological system modeling, electrogastrography, magnetogastrography