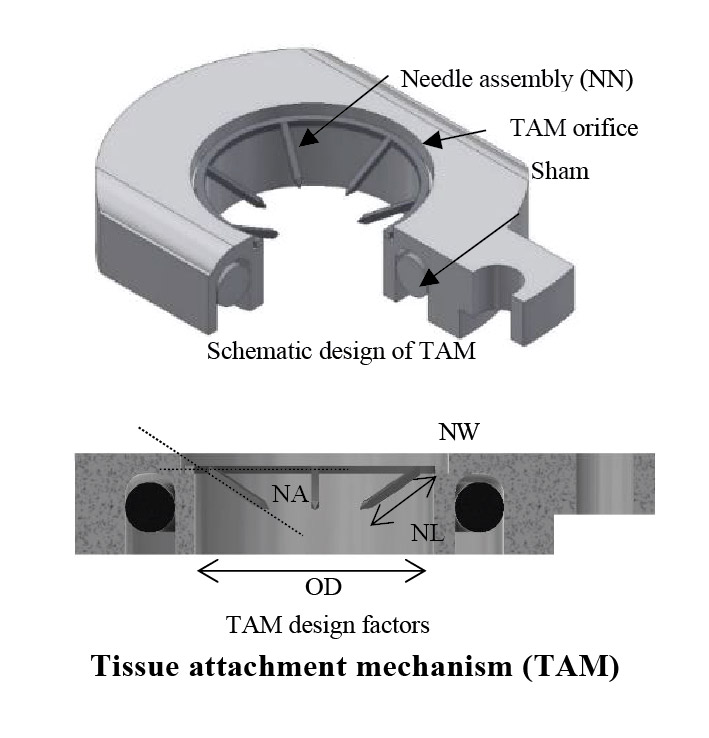
Biological macromolecule drugs or biologics are not suited for commonly preferred oral delivery due to intrinsic instability and physical, chemical, or immunological barriers to the gastrointestinal tract. Ingestible capsule robots (ICR) have become a versatile platform, including drug delivery applications for various gastrointestinal pathologies. In this work, a tissue attachment mechanism (TAM) for a drug delivery ICR was introduced to facilitate a non-invasive systemic delivery of unaltered biologics via direct injection through the insensate layers of the small intestine. The drug delivery ICR is designed so that it passively travels to the small intestine via digestive peristalsis after it is swallowed. Upon reaching the small intestine, the ICR deploys the drug-carrying payload onto the mucosa via the TAM. The TAM is then separated from the ICR and remains adhered to the mucosa for an extended period while maintaining intimate contact with the tissue. One potential application enabled by this prolonged intimate contact with tissue is extended-release drug delivery.
The main prerequisite for achieving systemic drug delivery via this device was to have a strong tissue attachment of the TAM. This study aimed to optimize the attachment success rate for drug delivery and characterize attachment duration in vivo. A fractional factorial approach was used in vivo to identify and optimize factors that most influence attachment of the TAM to maximize attachment rate. Optimization experiments were performed at multiple levels using the SI of anesthetized pigs, and an attachment success rate of 92% was achieved. Optimal TAMs were surgically placed in vivo to determine the duration of attachment following anesthetization and surgery recovery. The average attachment duration was found to be 32.2±9.4 hours. This work demonstrated a device for consistent and reliable attachment duration, making the TAM a suitable candidate for a 24-hour systemic drug delivery platform.