Survivors may face a slew of invisible health problems. Can a new software help?
At the start of the COVID-19 pandemic, doctors and hospitals were overburdened from patients suffering from the immediate, acute effects of the virus. But as time went on, it turned out that even the patients who survived—or had mild symptoms and didn’t require hospitalization—weren’t fully recovered.
In the largest study of COVID survivors, researchers found that seven months after infection, many individuals continued to suffer from a range of health conditions—many invisible—including but not limited to: malaise, fatigue, brain fog, vision problems, and more [1]. In addition, SARS-CoV-2—the virus that causes COVID-19—attacks the vasculature, which means the virus can affect blood supply to multiple organs, leading to more widespread problems. It’s common for these survivors to undergo extensive medical workups involving multiple scans and specialists, without any conclusive results, says Aaron Bunnell, medical director of rehabilitation consults at Harborview Medical Center in Seattle and co-medical director of the Post-COVID-19 Rehabilitation and Recovery Clinic at University of Washington’s School of Medicine. Not to mention, it can take weeks to complete all these tests.
About a year ago, UK-based digital health company Perspectum began working on a new software that interprets scans from magnetic resonance images (MRI) to better treat COVID survivors. This technology, called Coverscan, takes an MRI scan of organs that might be affected such as the heart, liver, lung, kidneys, spleen, and pancreas. It’s a thirty-five minute test that Perspectum hopes can help point COVID survivors to get the proper care [2]. “It’s a balanced scorecard of the body’s organs to holistically assess health,” says CEO Rajarshi Banerjee.
And the technology behind it may be promising for helping patients. “Using multiparametric scans and combining different sequences of MRI might give more sensitivity for organ damage,” says Bunnell, who is not involved with Perspectum. “That may be of interest because it may point us to where pathology is occurring and allow for more scientific investigation.” In other words, Coverscan might not only be a tool for patients, but also for researchers studying the impact of the virus on COVID survivors.
Coverscan relies on quantitative MRI, which means that instead of making medical conclusions from just the image, the software examines the characteristics of the MRI. (Patients don’t have to drink a contrast liquid, which some MRIs require to get detailed images.) Perspectum has developed algorithms and calculations that process those characteristics. The metrics that Coverscan measures are compared to what’s considered to be healthy and normal from rigorous data and previous research.
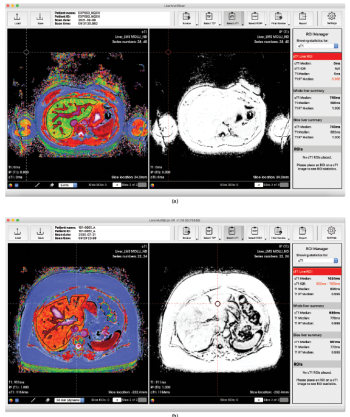
“Instead of looking at your hand, and saying that it’s five centimeters by four centimeters, it would say how much muscle or fat there is, or how much inflammation there is,” says Banerjee. For each organ, then, Coverscan will not just give clinicians the dimensions, but a measure of that organ’s health (Figure 1).
Good News for Diagnostics
Late last spring, Perspectum first wanted to study the impact of COVID-19 on individuals who survived—Banjeree thought they would produce a “good news story.” At the time, Perspectum had already developed quantitative diagnostic tools for liver disease and cancer. To develop Coverscan, he and his colleagues applied the same principle to other organs. They piloted Coverscan on about four dozen patients and Banerjee was immediately concerned: about a quarter of those individuals showed significant cardiovascular issues, he said, which seemed out of the ordinary.
Perspectum went on to survey more COVID survivors, and published an update in March in BMJ Open, which looked at 201 people [3]. Early data from this relatively small cohort showed that 60% of survivors had severe symptoms, ranging from myocarditis (inflammation of the heart) and impairment of other organs.
So far, Coverscan has been a benefit to patients who have enrolled in its ongoing clinical trial in the United Kingdom. Lyth Hishmeh, a COVID survivor based in the U.K. and the co-founder of LongCovidSOS, said that without Coverscan, or a way to decide what was potentially wrong with him, he was being dismissed by healthcare providers. “Doctors were just telling us, oh, you’re all anxious. There’s nothing wrong with you,” Hishmeh said. That ultimately weakens the patient’s trust in medical providers, he says, when the patient’s lived experience runs counter to a doctor’s judgement.
Hishmeh enrolled in the Coverscan trial, and the software was able to determine that he had a mild cardiac impairment, along with a swollen left kidney. He then was able to take the results of this scan to his provider so he could get targeted care and follow-up treatment. And the earlier a treatable issue can get detected, the better, says Amitava Banerjee, an associate professor in clinical data science at University College London (UCL), who is not related to Banerjee of Perspectum.
Not only can Coverscan help patients get targeted care quickly, but it also saves on cost. In the U.K., one Coverscan will run about US$1000, estimates Banerjee of Perspectum. “It isn’t that [cost] prohibitive,” he says, “because you’re saving from [using] four or five different scans.” Coverscan has already been cleared for use in the UK, and as of publication, is still under review by the U.S. Food and Drug Administration for its breakthrough device designation.
One current limitation of the scanning software, however, is that it can’t yet image all the organs that are suspected to be impacted by the damage caused by the virus. For instance, brain fog, a common symptom listed by survivors, can’t yet be detected by Coverscan, says Banerjee of Perspectum. And another challenge is that even if Coverscan detects an issue, the software can’t necessarily advise medical professionals on how to treat it. Brain fog is tricky, Banerjee says, because even if he had a way to detect it, there is no known treatment for it yet.
For now, scientists and clinicians believe that while there is promise for Coverscan, more research still needs to be done. Even though Coverscan can now identify whether or not patients have severe organ dysfunction from the long-term events of COVID-19, the software should still be further studied in clinical trials for accuracy. Banerjee of UCL argues that continually using Coverscan in the clinic can also be helpful in defining what long-COVID looks like clinically. To date, there is still no consensus on how post-COVID syndrome should be defined.
But patients can continue to benefit from its use, depending on when different health regulatory agencies across the world approve it for use. For Hishmeh, Coverscan gave him a piece of mind, and he hopes it can provide the same for others.
References
- R. Wachter, The Digital Doctor: Hope, Hype, and Harm at the Dawn of Medicine’s Computer Age. New York, NY, USA: McGraw-Hill, 2017.
- P. Barr, G. Elwyn, and I. Scholl, “Achieving patient engagement through shared decision-making,’’ in The Wiley Handbook of Healthcare Treatment Engagement: Theory, Research, and Clinical Practice, A. Hadler, S. Sutton, and L. Osterberg, Eds. Hoboken, NJ, USA: Wiley, 2020, ch. 28, pp. 531–550.
- J. S. Hirsch et al., “HARVEST, a longitudinal patient record summarizer,’’ J. Amer. Med. Inform. Assoc., vol. 22, no. 2, pp. 263–274, Mar. 2015.
- HARVEST: Longitudinal Patient Record Summarization. Accessed: Apr. 22, 2021. [Online]. Available: https://people.dbmi.columbia.edu/noemie/harvest/
- Centers for Medicare & Medicaid Services. National Health Expenditure Projections 2018–2027. Accessed: Apr. 21, 2021. [Online]. Available: https://www.cms.gov/Research-Statistics-Data-and-Systems/Statistics-Trends-and-Reports/NationalHealthExpendData/Downloads/ForecastSummary.pdf