Dr. Lihong V. Wang from Washington University in St. Louis recently published an article titled Photoacoustic tomography (PAT) on the renowned online encyclopedia Scholarpedia. In this article, he thoroughly reviewed the principle, development and applications of a fast-developing biomedical imaging modality – the photoacoustic (PA, sometimes called optoacoustic) imaging technique.
PA imaging is based on photoacoustic effect. In PA imaging, an ultra-short pulsed laser (photonic energy), with power restricted below the safety limit, is used to produce ultrasound (mechanical energy) in soft tissue caused by molecular vibrations due to transient thermal absorption. Inner structure is visualized by reconstructing the initial pressure distribution in the object. It is an imaging modality that relies on listening to the “sound” generated by “light.” Because scattering of ultrasound in biological bodies is 100-1000 times less than that of photons, optical contrast, high-resolution and deep penetration depth compared with other optical imaging modalities are combined by this technique.
Despite the fact that the PA effect was discovered by the legendary scientist Alexander G. Bell in the 1880s, its application to biomedicine was not achieved until a century later, in the 1980s, after reliable and intense optical sources were developed. It has been intensively studied ever since. PA imaging can be classified into two primary categories, namely the PA computed photoacoustic tomography (PACT) and 3D scanning tomography (PA microscopy, or PAM). Whereas the former relies on inverse algorithms to reconstruct internal structure with sensed acoustic signals, the latter takes advantage of A-scan-type depth visualization at different lateral locations to generate B-mode and C-scan mode volumetric views.
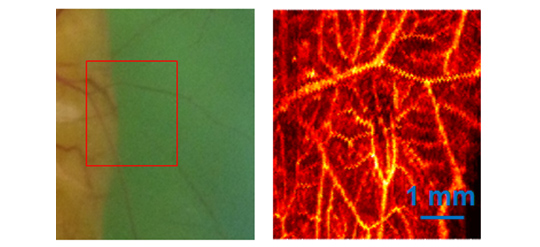
Optical absorption provides the contrast for PA imaging. Both endogenous (hemoglobin, melanin, DNA, RNA etc.) and exogenous absorbers can be imaged with this technique. As hemoglobin is the dominant absorber in biological bodies, PA imaging with multiple-wavelength pulsed-laser can reveal oxygen content in soft tissue (functional information). PA imaging is the only available imaging modality that is capable of providing high-resolution “structural, functional and molecular imaging of organelles, cells, tissues, and organs in vivo.”
Applications of PA imaging are being intensively studied in a large number of directions, including fundamental, preclinical and clinical studies. According to this article, examples of preclinical studies include:
- Non-fluorescent pigments (red blood cells & melanin)
- Angiogenesis and anti-angiogenic response
- Microcirculation physiology and pathology
- Drug response for screening
- Brain functions
- Biomarkers
- Gene activities through reporter genes
Clinical studies include:
- Melanoma cancer screening
- Gastrointestinal tract endoscopy
- Intravascular catheter imaging
- Neonatal and adult brain imaging
- Breast cancer detection
- Prostate cancer detection
- Guided sentinel lymph node needle/core biopsy for breast cancer staging
- Early response to chemotherapy
- Dosimetry in thermal therapy
- In vivo label-free histology by photoacoustic imaging of cell nuclei
- Blood flow, oxygenation, and tissue metabolism imaging”
The main limitation of PA imaging is the limited penetration depth when high resolution is desired. For a resolution of < 1 mm, only 5 ~ 7 cm-penetration can be achieved. Optical resolution can be achieved with a penetration depth of only ~ 1 mm. Nevertheless, PA imaging still provides excellent penetration-to-resolution ratio. For instance, the number is over 100 in acoustic-resolution photoacoustic microscopy. Other challenges include acoustic energy coupling in the sensing process and imaging speed while dealing with pulsed laser.
Besides the Optical Imaging Lab directed by Dr. Lihong V. Wang at Washington University in St. Louis, which pioneered research on PA imaging, there are a number of major groups working in this field. One example is the Photoacoustic Imaging Group with the Department of Biophysics and Bioengineering at University College London in the U.K. Solid work has been accomplished by this group encompassing “the full range of activities” in PA imaging, from instrumentation, image reconstruction algorithms, and image processing to preclinical studies. Studies are also being conducted by quite a number of other labs distributing in North America, Europe and Asia. In late 2013, an open-access journal Photoacoustics was launched by Elsevier, for which Prof. Vasilis Ntziachristos serves as the Editor-in-Chief. The scope of this journal is described as “to publish original research and review contributions within the fast growing field of photoacoustics (optoacoustics) and thermoacoustics.”
Commercialization of the PA technique has also been launched with products for preclinical and basic research at different locations globally, examples of which include TomoWave Laboratories, Inc. and Endra Life Sciences in the U.S., iThera Medical GmbH in Germany specializing in PACT (Multispectral Optoacoustic Tomography), and FUJIFILM VisualSonics, Inc. located in Canada providing scanning PAT imaging products based on a high-frequency, high-resolution B-mode ultrasound imaging system.
Studies on PA imaging have been booming in the last two decades, due to its capability for multi-scale, high-resolution, non-invasive imaging of both structural and functional information. It is now migrating fast toward preclinical and clinical studies and is anticipated to be a powerful tool for both fundamental and clinical studies.
References
- Lihong Wang (2014), Scholarpedia, 9(2):10278.
- M. Xu and L. V. Wang (2006), “Photoacoustic imaging in biomedicine,” Review of Scientific Instruments 77, 041101-(1–22).
- Wang, L. V. (2009). “Multiscale photoacoustic microscopy and computed tomography.” Nature Photonics 3: 503-509.
- Laufer J, Johnson P, Zhang E, Treeby B, Cox B, Pedley B, Beard P (2012) In vivo preclinical photoacoustic imaging of tumor vasculature development and therapy, Journal of Biomedical Optics 17(5), 056016.
- http://www.medphys.ucl.ac.uk/research/mle/index.htm
